Enhanced enzymatic activity exerted by a packed assembly of single type of enzyme
Research Topics / 研究トピックス
An article written by H. Dinh, E. Nakata, K. Mutsuda-Zapater, M. Saimura, M. Kinoshita, T. Morii was published in Chemical Science.
Title; Enhanced enzymatic activity exerted by a packed assembly of single type of enzyme
DOI: 10.1039/D0SC03498C
This article is part of the themed collection: 2020 Chemical Science HOT Article Collection
Advanced Energy Utilization Research Division, Biofunctional Chemistry Section.
2020.July 27
Advanced Energy Conversion Division Biofunctional Chemistry Research Section
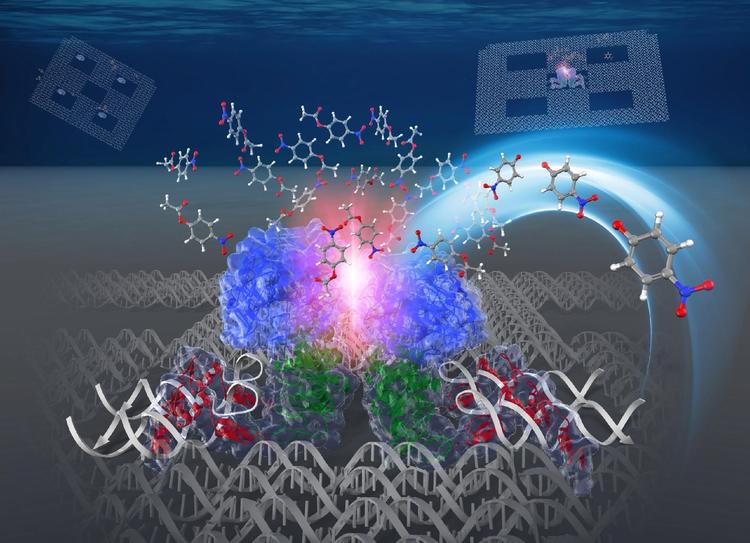
In contrast to the dilute conditions employed for in vitro biochemical studies, enzymes are spatially organized at high density in cellular micro-compartments. A single type of monomeric enzyme carbonic anhydrase was located on a DNA scaffold in the packed or dispersed states. At the same concentration of enzyme, reaction proceeded faster in the packed than in the dispersed state. We propose that the entropic force of water increases local substrate concentration within the domain confined between enzyme surfaces, thus accelerating the reaction.








