Self-Assembly Science
The Laboratory for Complex Energy Processes Self-Assembly Science Research Section
Professor (Concurrent): Masato KATAHIRA
The aim of this research is to construct the supramolecular assemblies of the topologically interlocked components inside a DNA origami. Such assemblies of the functional structures are promising in the fields of molecular switches, motors, sensors, and logic devices.
Nanomolecular fabrication of supramolecular assemblies
DNA molecules are not merely associated with genetics and the carrying of information. They have been used as excellent construction units in structural DNA nanotechnology due to their unique structural motifs and robust physicochemical properties. I have been working on the self-assembly of DNA origami (a method to create nanostructures by folding DNA) nanostructures to create micrometer scale structures that can be used for several applications such as fabrication of nanodevices, analysis of biomolecular reactions, and templates for various applications. Also, I have utilized these nanostructures for the single molecule analysis of various biomolecular reactions, structure and function of DNA and proteins, and enzymes related to biomass energy conversion.
Recently, I have been collaborating with the research groups of Dr. Eiji Nakata (IAE, Kyoto University) and Prof. Youngjoo Kwon (Ewha Womans University) for the nanofabrication of the topologically interlocked supramolecular assemblies. Topologically interesting structures such as Borromean rings, catenanes, rotaxanes, and knots have been prepared by using duplex DNAs. Also, the complexity of the catenane and rotaxane structures were increased by constructing them by the DNA origami method. However, integration of the duplex DNA catenanes and rotaxanes with functional sequences to the relatively larger and complex DNA nanostructures such as DNA origami has not yet been realized. We have successfully fabricated the DNA catenane and rotaxane structures inside a frame-shaped DNA origami. Apart from the applications in nanotechnology, these interlocked structures can be used for the biomolecular analysis, such as enzymatic reactions and drug screening. For example, these topological structures can be used as the potential substrates for the topoisomerase (Topos) enzymes, and screening of Topo inhibitors.
Among the various types of DNA-binding proteins, Topos are quite attractive due to their importance in cancer therapy. Topos regulate the topological problems of DNA that arises due to the intertwined nature of the double helical structure. These enzymes also play an important role in various biological processes such as replication, transcription, recombination, and chromosome condensation and segregation. Topos resolve the topological problems by transiently cleaving the phosphodiester bond, which generates a Topo-DNA cleavage complex.
Once the winding stress is resolved, the Topo-mediated DNA break is resealed. This process is critical for the healthy cells to survive and function normally. Failure to reseal the DNA break can ultimately lead to cell death. This Topo-DNA cleavage complex and various other steps (such as binding of Topo to DNA, ATP driven strand passage, strand cleavage by Topo, formation of Topo-DNA cleavage complex, religation of cleaved DNA, and catalytic cycle after DNA cleavage/enzyme turnover) involved in the Topos function are of great interest as potential targets for the development of anticancer drugs. Despite the development of various Topo-inhibitors, the mechanism of action of these anticancer drug molecules is not well known. Thus, to understand the Topos reaction and the mechanism of the inhibitors, it is necessary to develop an elegant method.
Here, we aim to develop a novel method by using our supramolecular assemblies of the catenane and rotaxane inside a DNA origami and high-speed atomic force microscopy (HSAFM) for the screening of Topo-inhibitors. The formation of the DNA origami frame and the insertion of the catenane and rotaxane structures were characterized. The Topo reactions and the function of Topo-inhibitors are under investigation. Apart from the Topo reactions and inhibitor screening, the supramolecular assemblies of the topologically interlocked components inside a DNA origami are also promising in the fields of molecular switches, motors, sensors, and logic devices.
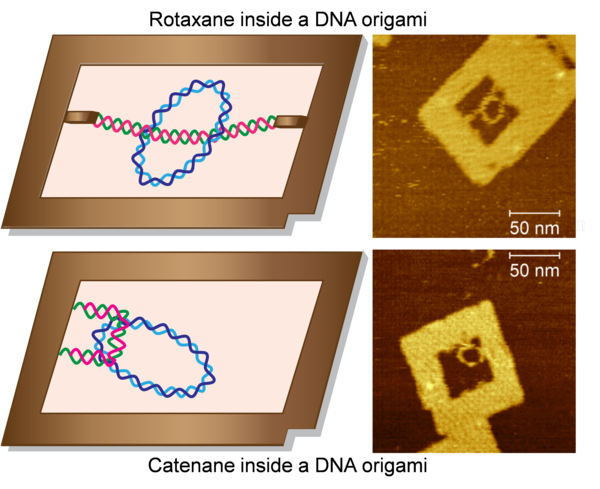 DNA rotaxane and catenane inside a DNA origami frame Left: The illustration of the topologically interlocked DNA rotaxane and catenane inside a DNA origami frame. Right: AFM images of the respective structures
DNA rotaxane and catenane inside a DNA origami frame Left: The illustration of the topologically interlocked DNA rotaxane and catenane inside a DNA origami frame. Right: AFM images of the respective structures








